 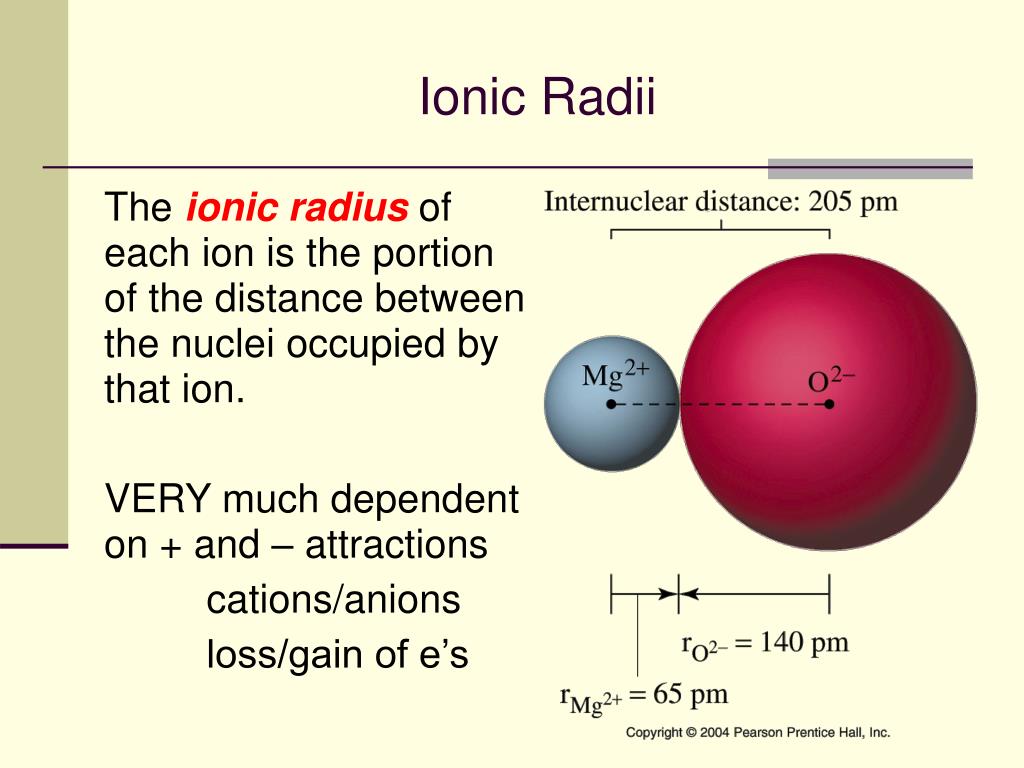
These are ionic radius, covalent radius, van der Waals radius, etc and all these properties have the same trend as the atomic radius. In a silicon atom, the force of attraction from the nucleus to the outer energy level is. There are many types of the radius of the element that explains the distance of the electrons. nitrogen has bigger charges a greater attraction. Therefore, the correct answer is option (a). Since both of them belong to the same period, the magnesium will be smaller due to the smaller effective nuclear charge. Answer: Cadmium has larger atomic radius than sulfur. Now, magnesium is the element of the s-block having atomic number 12 and belongs to period three and phosphorus is the element of the p-block having atomic number 15 and also belongs to the third period. When we move down the group in the periodic table, the atomic radius increases because the number of shells increases but the electrons in the shell remain the same due to which the nuclear charge decreases and the distance of the outermost electron increases. When we move left to right in the period of the periodic table, the atomic radius decreases because the number of electrons increases but the number of shells remains the same due to which the nuclear charge increases and the electrons get attracted towards the nucleus. In the periodic table, there is a trend in the atomic radius of the elements. The atomic radius of the atom is explained as the distance between the nucleus of the atom and the outermost electron of the element. Does oxygen have a greatest atomic radius then. The atomic size of the elements can be explained by the factor of the nuclear charge. Which element has a larger atomic radius than sulfur Many elements have a larger atomic radius than sulfur (100 pm): selenium, tellurium, uranium. Metals are placed on the left side of the periodic table and the nonmetals are placed on the right side of the periodic table. Hint: The metals have a larger atomic radius than the non-metals. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
